Western blotting is commonly used to determine the presence of a certain protein in a sample. Gel electrophoresis is used to separate the proteins in the first stage. The isolated protein is then put to the PVDF membrane's nitrocellulose layer and analyzed with particular antibodies.
The Western blot reagent used for this purpose is essential for successful absorption experiments. This technique has a wide range of applications in various fields of molecular biology. You can get more information on western blot from various sites like www.bosterbio.com/services/assay-services/western-blotting-service.
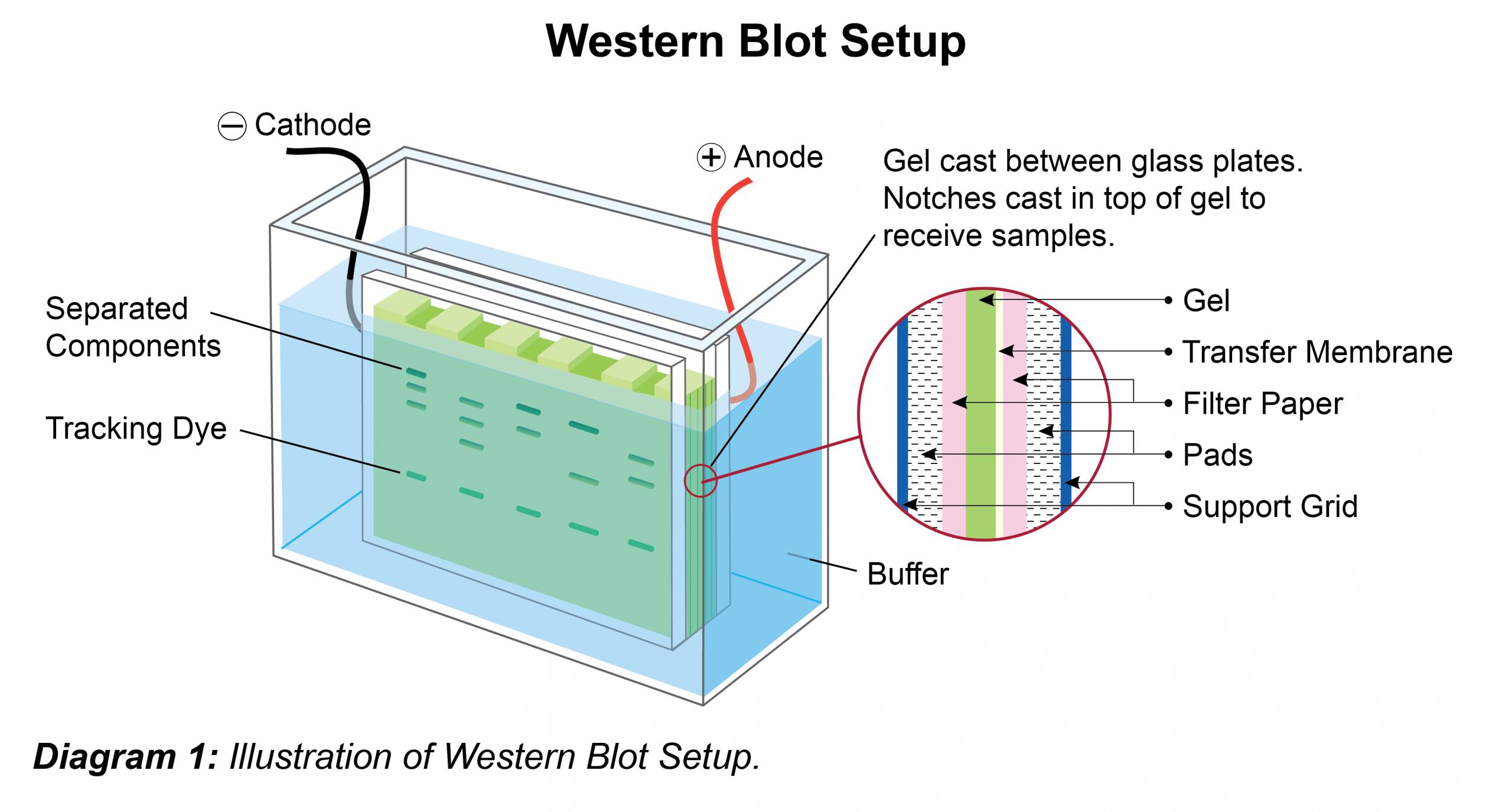
Image Source: Google
In conventional absorption methods, there are two steps to detecting protein after gel electrophoresis. The protein is first incubated with the primary antibody solution. Second, the bound primary antibody is washed and treated with a more specific secondary antibody.
Often, secondary antibodies can bind to primary antibodies and increase signal intensity for better resolution. The most commonly used secondary antibodies produce luminescence because they are conjugated to horseradish peroxidase, which produces a signal proportional to the amount of protein in the sample.
The light from this reaction can be used to sensitize photographic film when it is held against the membrane to obtain a paper copy of the smudge. Point analysis was carried out after washing unused probes. The points are then compared with the markers for a better estimate.
Care must be taken to avoid errors during analysis. In western spot colorimetric detection, the intensity of staining is measured using spectrophotometry or densitometry to determine the amount of protein.